A HosCom International 2025 Vol. 2 Article
World Information
The Role of Decolonization Strategies in Preventing Healthcare-Associated Infection (HAIs)
Authors:
Professor. Sasheela Ponnampalavanar
Infectious Disease Consultant, Faculty of Medicine, University of Malaya, Malaysia
The Landscape of Healthcare-Associated Infections (HAIs)
The pathogenesis of HAIs involves complex interactions between the host, the pathogen, and the healthcare environment. Patients in hospitals, particularly those who are immunocompromised or undergoing invasive procedures, are at increased risk of acquiring infections.
Colonization refers to the presence of bacteria on the body without causing disease. In hospitals, patients can be colonized or infected with both multidrug-resistant organisms (MDROs) and non-MDRO pathogens. Patient secretions and excretions can contaminate their surroundings, including surfaces and medical equipment. These pathogens can then be transmitted to susceptible patients via direct contact ( e.g., healthcare workers' hands) or indirect contact (e.g., contaminated surfaces or shared equipment) (Figure). MDRO patients and their environments are both a reservoir and a source of further transmission. Various studies have shown that patients with MDRO contaminate their environments (Lerner et al.,2023; Schechner et al., 2023; Chia et al., 2020). A systematic review published in the Journal of Hospital Infection (Gu et al., 2023) shows that the exposure of bed occupants to infected or colonized MDROs significantly increases the risk of MDRO acquisition in subsequent bed occupants. The use of antimicrobials further complicates this dynamic by selecting for resistant organisms, altering the patient's microbiome, and potentially facilitating the emergence of new MDROs.
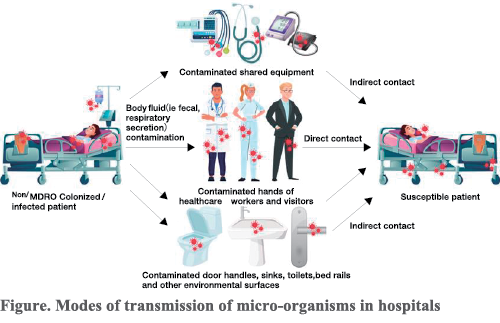
Once a patient is colonized with a pathogen, they are at increased risk of HAIs, which arise through two distinct pathways. Endogenous infections occur when an infection is caused by the patient's own colonizing organisms, which can include normal flora or organisms acquired during the hospital stay, and become invasive under conducive conditions. For example, the cumulative incidence of infection after 30 days of colonization with vancomycin-resistant enterococci (VRE) was 8,% and multidrug-resistant Gram-negative rods was 14% (Septimus & Schweizer, 2016). Patients who had nasal colonization with Staphylococcus aureus were 2-4 times more likely than others to develop an infection. (Honda et al., 2010). Exogenous infections result from pathogens introduced into the patient's body from the hospital environment, such as through contaminated devices, healthcare workers' hands, or surfaces.
Decolonization Strategies
Considering that colonized patients can act as reservoirs for the transmission of pathogens and are at an increased risk for infection, decolonization plays a significant role in infection prevention.
Decolonization refers to any intervention that eliminates detectable pathogen carriage from any site on the body. The primary goals of decolonization are to reduce the bioburden on body surfaces, thereby minimizing environmental contamination, subsequent transmission, and exogenous infections. Additionally, decolonization lowers the risk of endogenous infections, particularly in cases where skin integrity is compromised due to the use of medical devices (Edmiston et al., 2008; Rhee et al., 2018).
Universal decolonization (horizontal strategies) involves applying decolonization measures to all patients, especially in settings where co-colonization with multiple MDROs is common- such as in ICUs or among patients with medical devices in situ. This typically includes skin decolonization, with or without nasal decolonization.
Targeted decolonization (vertical strategies) involves active screening for specific pathogens followed by targeted interventions, such as skin decolonization and nasal decolonization for S. aureus. Skin decolonization typically involves the use of antiseptics like chlorhexidine gluconate (CHG). The contact time of application is crucial for maximizing the reduction of microbial load and achieving successful decolonization. Nasal decolonization is commonly achieved using mupirocin ointment or povidone-iodine to eradicate the nasal passages.
Effectiveness of Decolonization in Infection Prevention
The efficacy of decolonization strategies varies depending on several factors, including the type of pathogen, the site of colonization, and adherence to decolonization protocols.
Numerous studies have demonstrated the effectiveness of CHG bathing in reducing the incidence ofHAis, particularly in high-risk patient populations. A prospective, sequential-group, single-arm clinical trial in an ICU by Vernon et al. (2006) found that cleansing patients with chlorhexidine-saturated cloths reduced VRE contamination of patients' skin (2.5 log reduction), the environment (34% to 11 %), and health care workers' hands (56% to 37%), in addition to decreasing patient acquisition ofVRE (20% to 8%).
Systematic reviews have highlighted the efficacy of CHG bathing in reducing hospital-acquired bloodstream infections (HA-BSI), VRE carriage, and incidences of CLABSI, VAP, CAUTI, and SSI (Derde et al., 2014; Denny & Munro, 2016; Huang et al., 2016; Musuuza et al., 2019). Although there is little evidence in these reviews for the efficacy of CHG bathing against multidrug-resistant gram-negative bacteria (MDR GNB), quasi-experimental studies have shown a significant reduction in MDR GNB colonization after CHG bathing, including carbapenem-resistant enterobacterales (CRE) and carbapenem-resistant Acinetobacter baumani (CRAB) (Borer et al., 2007; Abboud et al., 2016; Apisarnthanarak et al., 2014a; Ruiz et al., 2017). Many of these studies were conducted in endemic settings, especially ICUs. Of note, the impact of decolonization in decreasing gram-negative multi-drug resistant organism (GN MDRO) was fouod to be greater if the hospital wards had higher rates at baseline and if other interventions were used concurrently, such as hand hygiene, environmental cleaning, contact precautions, and education (Ruiz et al., 2017; Gall et al., 2020).
With regard to whether universal or targeted decolonization is better, a cluster randomized controlled trial (RCT) (REDUCE MRSA Trial) in the United States found that routine universal decolonization in ICUs was more effective than targeted decolonization or screening and isolation in reducing rates of methicillin-resistant S. aureus (MRSA) clinical isolates (33% reduction) and bloodstream infection from any pathogen (44% reduction) (Huang et al., 2013).
The impact of decolonization is not well studied in patients outside of the ICU. The ABATE cluster RCT showed that, although decolonization with universal CHG bathing and targeted mupirocin for MRSA carriers did not significantly reduce multidrug-resistant organisms in non-critical-care patients, it was effective in reducing MRSA, VRE, and all-cause bloodstream infection in patients with medical devices (Huang et al., 2019).
Use of CHG also generates substantial cost savings for healthcare facilities. Dixon and Carver (20 I 0) reported that, when compared to standard soap and water, the introduction of CHG bathing in a nine-bed surgical ICU led to a cost savings of $728,820 over a 17-month period.
Challenges and Considerations
One of the most significant challenges associated with decolonization is the potential for promoting antimicrobial resistance. In a study by Apisamthanarak et al. at a Thai hospital, there was a correlation between CHG consumption and an increase in Acinetobacter baumannii chlorhexidine MIC, although it did not achieve the threshold for the emergence of chlorhexidine-resistant extensively drug-resistant (XDR) A. baumannii detection. Furthermore, the incidence of XDR A. baumannii did not increase across hospital units or specimen sources in this study (Apisarnthanarak et al., 2014b). In the United States, where CHG decolonization strategies have been implemented, MRSA isolates with increased MICs and/or qacA/B genes have seldom been reported (<1%) despite increasing CHG use (Babiker et al., 2021 ). Therefore, the concern about resistance should not hinder the use of CHG, especially given the substantial evidence supporting its effectiveness in preventing HAIs. However, it is essential to maintain ongoing and active surveillance for chlorhexidine-resistant pathogens in order to monitor and evaluate the potential emergence of resistance. This will ensure that decolonization strategies remain both effective and safe in preventing HAIs.
The effectiveness of decolonization relies on the correct application of CHG, particularly ensuring adequate contact time as well as strict adherence to established protocols. Without strict compliance, the benefits of decolonization efforts can be significantly diminished, highlighting the need for continuous training, education, and monitoring.
Conclusion
Decolonization plays a critical role in infection prevention, particularly in high-risk populations such as ICU patients, those with medical devices, or individuals colonized with MDROs. As the healthcare landscape continues to evolve, ongoing research and innovation are essential to refine decolonization strategies and optimize their effectiveness in preventing infections. By integrating decolonization with other infection prevention measures, such as antimicrobial stewardship, environmental cleaning, and hand hygiene, healthcare facilities can create a comprehensive approach to reducing the burden of HAIs and controlling outbreaks, thereby safeguarding the health of both patients and healthcare workers.
-
Publication Date:November 26, 2025
-
Category:HAI
HosCom International 2025 Vol. 2

Other Articles in this volume
References
- Abboud, C. S., de Souza, E. E., Zandonadi, E. C., Borges, L. S., Miglioli, L, Monaco, F. C., Barbosa, V. L., Cortez, D., Bianco, A C., Braz, A, & Monteiro, J. (2016). Carbapenemresistant Enterobacteriaceae on a cardiac surgery intensive care unit: Successful measures for infection control. Journal of Hospital Infection. 94(1), 60-64.
- Apisarnthanarak, A, Pinitchai, U., Warachan, B., Warren, D. K, Khawcharoenporn, T., & Hayden, M. K. (2014a). Effectiveness of infection prevention measures featuring advanced source control and environmental cleaning to limit transmission of extremely drug-resistant Acinetobacter baumannii in a Thai intensive care unit: An analysis before and after extensive flooding. American Journal of Infection Control, 42(2), 116-- 121. https://doi.org/10.1016/j.ajic.2013.09.025
- Apisarnthanarak, A., Hsu, L. Y.; Lim, T.-P., & Mundy, L. M. (2014b). Increase in chlorhexidine minimal inhibitory concentration of Acinetobacter baumannii clinical isolates after implementation of advanced source control. Infection Control & Hospital Epidemiology, 35(1), 98--99. https://doi.org/10.1086/674404
- Babiker, Ahmed, et al. Assessing the potential for unintended microbial consequences of routine chlorhexidine bathing for prevention of healthcare-associated infections. Clinical Infectious Diseases 72.5 (2021): 891-898.
- Borer, A, Gilad, J., Porat, N., Megrelesvilli, R., Saidel-Odes, L., Peled, N., Eskira, S., Schlaeffer, F., & Almog, Y. (2007). Impact of 4% chlorhexidine whole-body washing on multidrug-resistant Acinetobacter baumannii skin colonisation among patients in a medical intensive care unit. Journal of Hospital Infection, 67(2), 149--155. https://doi.org/10.1016/j.jhin.2007.07.023
- Chia, P. Y., Sengupta, S., Kukreja, A., Ponnampalavanar, S.S. L, Ng, 0. T., & Marimuthu, K (2020). The role of hospital environment in transmissions of multidrug-resistant gram-negative organisms. Antimicrobial Resistance & Infection Control, 9(29). https://doi.org/10.1186/s13756-020-0685-l
- Denny, J., & Munro, C. L. (2016). Chlorhexidine bathing effects on healthcare-associated infections. Biological Research for Nursing, 19(2), 123-136. https://doi.org/10.1177/1099800416654013
- Derde, L. P. G., Cooper, B. S., Goossens, H., Malhotra-Kumar, S., Willems, R. J. L., Gniadkowski, M., Hryniewicz, W., Empel, J., Dautzenberg, M. J. D., Almane, D., Aragao, I., Chalfine, A., Dumpis, U., Esteves, F., Giamarellou, H., Muzlovic, I., Nardi, G., Petrikkos, G. L, Tomic, V., & Marti, AT. (2014). Interventions to reduce colonisation and transmission of antimicrobial-resistant bacteria in intensive care units: An interrupted time series study and cluster randomised trial. The Lancet Infectious Diseases. 14(1), 31-39. https://doi.org/10.1016/s1473-3099(13)70295-0
- Dixon, J.M., & Carver, R. L. (2010). Daily chlorohexidine gluconate bathing with impregnated cloths results in a statistically significant reduction in central line-associated bloodstream infections. American Journal of Infection Control, 38(10), 817-821. https://doi.org/10.1016/j.ajic.2010.06.005
- Edmiston, C. E., Krepel, C. J., Seabrook, G. R., Lewis, B. D., Brown, KR., & Towne, J.B. (2008). Preoperative shower revisited: Can high topical antiseptic levels be achieved on the skin surface before surgical admission? Journal of the American College of Surgeons, 207(2), 233-239. https://doi.org/10.1016/j.jamcollsurg.2007.12.054
- Gall, E., Long, A, & Hall, K K (2020). Chlorhexidine bathing strategies for multidrug resistant organisms: A summary of recent evidence. Journal of Patient Safety, 16(3), S16--S22. https://doi.org/10.1097/pts.0000000000000743
- Gu, G. Y., Chen, M., Pan, J. C., & Xiong, X. L. (2023). Risk of multi-drug-resistant organism acquisition from prior bed occupants in the intensive care unit: A metaanalysis. Journal of Hospital Infection, 139, 44-55. https://doi.org/10.1016/j.jhin.2023.06.020
- Honda H., Krauss M. J., Coopersmith C. M., Kollef, M. H., Richmond, AM. Fraser, V. J., & Warren, D. K (2010). Staphylococcus aureus nasal colonization and subsequent infection in intensive care unit patients: Does methicillin resistance matter? Infection Control & Hospital Epidemiology, 31(6), 584-591. https://doi.org/10.1086/652530
- Huang, H., Chen, B., Wang, H.-Y., & He, M. (2016). The efficacy of daily chlorhexidine bathing for preventing healthcare-associated infections in adult intensive care units. The Korean Journal of Internal Medicine, 31(6), 1159-1170. https://doi.org/10.3904/kjirn.2015.240
- Huang, S. S., Septirnus, E., Kleirnnan, K, Moody, J.. Hickok, J., Avery, T. R., Lankiewicz, J., Gombosev, A, Terpstra, L., Hartford, F., Hayden, M. K, Jernigan, J. A., Weinstein, RA, Fraser, V. J., Haffenreffer, K, Cui, E., Kaganov, R E., Lolans, K, Perlin, J. B., & Platt, R. (2013). Targeted versus universal decolonization to prevent ICU infection. The New England Journal of Medicine, 368(24), 2255-2265. https://doi.org/10.1056/nejmoa1207290
- Huang, S.S., Septimus, E., Kleinman, K, Moody, J., Hickok, J., Heim, L., Gombosev, A, Avery, T. R., Haffenreffer, K, Shirnelman, L, Hayden, M. K, Weinstein, R. A, Spencer-Smith, C., Kaganov, R. E., Murphy, M. V., Forehand, T., Lankiewicz, J., Coady, M. H., Portillo, L., & Sarnp-Patel, J. (2019). Chlorhexidine versus routine bathing to prevent multidrug-resistant organisms and all-cause bloodstream infections in general medical and surgical units (ABATE Infection trial): A cluster-randomised trial. The Lancet, 393(10177), 1205-1215. https://doi.org/10.1016/s0140-6736(18)32593-5
- Musuuza, J. S., Guru, P. K, O'Horo, J. C., Bongiorno, C. M., Korobkin, M. A, Gangnon, R. E., & Safdar, N. (2019). The impact of chlorhexidine bathing on hospital-acquired bloodstream infections: A systematic review and meta-analysis. BMC Infectious Diseases. 19(1). https://doi.org/10.1186/sl2879-019-4002-7
- Rhee, Y., Palmer, L. J., Okamoto, K, Gemunden, S., Harnrnouda, K., Kemble, S. K, Lin, M. Y., Lolans, K, Fogg, L., Guanaga, D., Yokoe, D.S., Weinstein, R. A, Frend!, G., Hayden, M. K., & Centers for Disease Control and Prevention Epicenter Program. (2018). Differential effects of chlorhexidine skin cleansing methods on residual chlorhexidine skin concentrations and bacterial recovery. Infection Control & Hospital Epidemiology, 39(4), 405--411. https://doi.org/10.1017/ice.2017.312
- Ruiz, J., Ramirez, P., Villarreal, E., Gordon, M., Saez, I., Rodríguez, A., Castafieda, M. J., & Castellanos-Ortega, A. (2017). Daily bathing strategies and cross-transmission of multidrug-resistant organisms: Impact of chlorhexidine-impregnated wipes in a multidrug-resistant gram-negative bacteria endemic intensive care unit. American Journal of Infection Control, 45(10), 1069-1073. https://doi.org/10.1016/j.ajic.2017.06.029
- Schechner, V., Lerner, A. 0., Temkin, E., & Carmeli, Y. (2023). Carbapenem-resistant Acinetobacter baumannii load in patients and their environment: The importance of detecting carriers. Infection Control & Hospital Epidemiology, 44(10), 1670-1672. https://doi.org/10.1017/ice.2023.39
- Septimus, E. J., & Schweizer, M. L (2016). Decolonization in prevention of healthcare associated infections. Clinical Microbiology Reviews, 29(2), 201-222. https://doi.org/10.1128/crnr.00049-15
- Vernon, M. 0., Hayden, M. K, Trick, W. E., Hayes, R. A, Blom, D. W., & Weinstein, R. A (2006). Chlorhexidine gluconate to cleanse patients in a medical intensive care unit. The effectiveness of source control to reduce the bio burden of vancomycin-resistant enterococci. Archives of Internal Medicine, 166(3), 306--312. https://doi.org/10.1001/archinte.166.3.306
- World Health Organization. (2023). Key Facts and Figures: World Hand Hygiene Day 2023. World Health Organization. www.who.int. https://www.who.int/campaigns/worldhand-hygiene-day/2023/key-facts-and-figures